|
11/24/2023 0 Comments Practice calculating formal charge  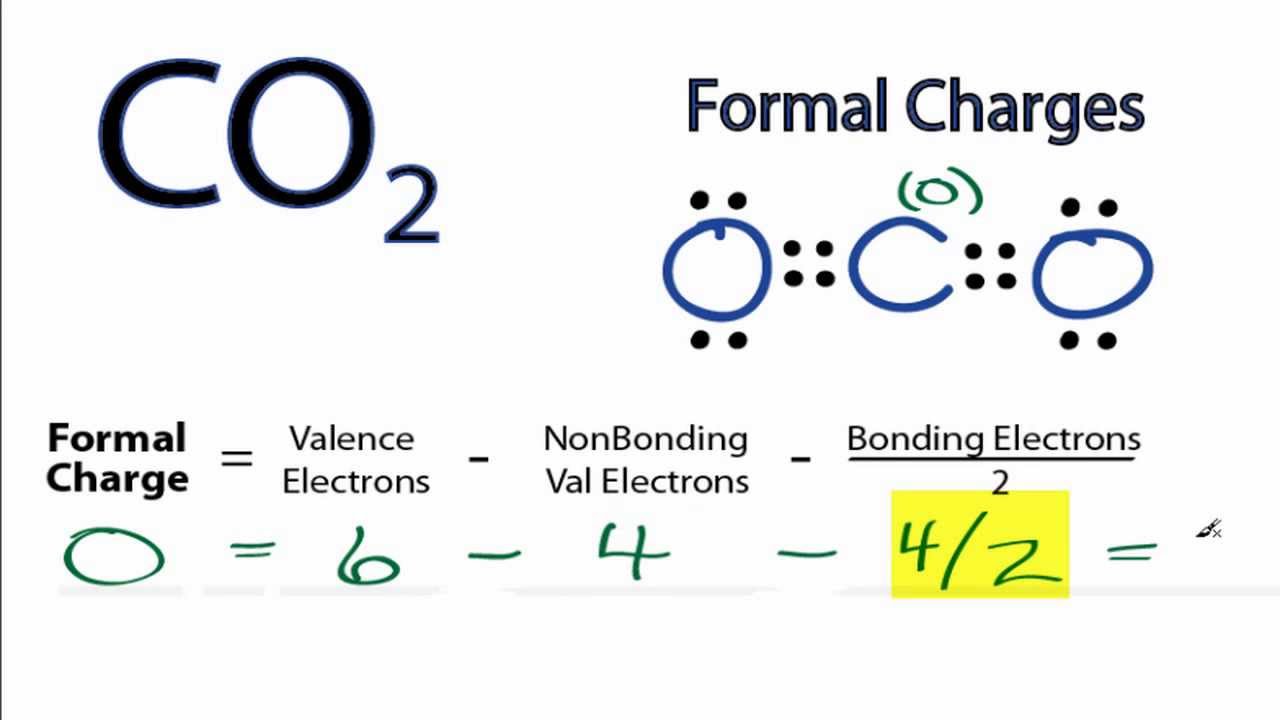
What that means is I'm gonna be looking at group numbers and I'm gonna be looking at sticks and thoughts. Let's go ahead and do this example where I wanna look at each atom and I want to count the formal charges for all of them. There's usually not that many formal charges on the molecule. You're just looking at each Adam and saying, Does this have a formal charge? Does this have a formal charge? The net charges the collection of all of those sums together. You see it a little bit confused over like, Oh, does the formal charge go on the whole thing? Or is it just one, Adam? No. Now, this is an important point because I remember when I was an undergrad. The net charge is the term that we give for the sum of all the formal charges. So you take your group number, then you just subtract the sticks and the dots and you're good. A lot of times, you'll just be able to do this on your fingers. And then you subtract the valence electrons, which is just the sticks and the dots. 
So all you do to calculate formal charge is you take the group number, whatever that is, that could be Group four, Group five, whatever. So remember that the group number is how maney it wants the valence electrons, the sticks and the dots are the it actually has. Basically, a formal charges assigned whenever there's a difference between the number of Valence electrons and Adam wants toe have and the number of valence electrons it actually has. So let's go ahead and just jump right into it. And formal charges are just based on the entire idea of bonding preferences. 
So now that we know the formal charge formula, click on to the next video on, Let's put it to practice with the given example.So now that we understand bonding preferences so well, I want to move to a really related topic called formal charges. This is the sum off all formal charges within a compound. We're going to say remember Valence electrons equals the group number off that element, and when we're talking about non bonding electrons, realized that we're gonna count them individually with formal charge when we find the formal charge of every element within a compound, we can calculate what's called the Net charge. We'd say formal charge equals the valence electrons off that element that we're examining minus the bonds that it is making, plus the non bonding electrons that it possesses. Bonding electrons are the electrons that do participate in bonding with other elements, and non bonding electrons are the electrons that don't participate and bonding with other elements with formal charge. We assume that all atoms that are sharing electrons between each other they share them equally now with formal charge were introduced to terms such as bonding, electrons versus non bonding electrons. But when it comes to formal charge, we don't take that into consideration. In reality, sometimes an element that's more electro negative will hold on to that electron within the electrons within the bond a little bit more tightly around themselves, which inflates or makes the electron clouds larger. Now formal charge represents the charge given toe elements when assuming electrons are shared equally, regardless off electoral negativity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
